How Do You Know if a Mokecuel Is Chiral or Achiral
Stereoisomers Two
Chirality and Symmetry
All objects may be classified with respect to a belongings we call chirality (from the Greek cheir significant hand). A chiral object is not identical in all respects (i.due east. superimposable) with its mirror image. An achiral object is identical with (superimposable on) its mirror prototype. Chiral objects have a "handedness", for example, golf clubs, scissors, shoes and a corkscrew. Thus, one can purchase right or left-handed golf clubs and scissors. As well, gloves and shoes come in pairs, a correct and a left. Achiral objects do non have a handedness, for example, a baseball game bat (no writing or logos on it), a plain round ball, a pencil, a T-shirt and a nail. The chirality of an object is related to its symmetry, and to this end information technology is useful to recognize certain symmetry elements that may be associated with a given object. A symmetry element is a plane, a line or a point in or through an object, well-nigh which a rotation or reflection leaves the object in an orientation indistinguishable from the original. Some examples of symmetry elements are shown beneath.
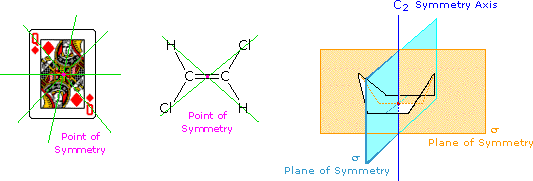
The face playing card provides an example of a centre or point of symmetry. Starting from such a indicate, a line fatigued in whatever direction encounters the same structural features as the opposite (180º) line. 4 random lines of this kind are shown in greenish. An example of a molecular configuration having a bespeak of symmetry is (E)-1,2-dichloroethene. Another way of describing a bespeak of symmetry is to annotation that any point in the object is reproduced past reflection through the center onto the other side. In these two cases the signal of symmetry is colored magenta.
The gunkhole conformation of cyclohexane shows an centrality of symmetry (labeled C2 here) and 2 intersecting planes of symmetry (labeled σ). The notation for a symmetry axis is Cdue north, where n is an integer chosen so that rotation about the axis past 360/nº returns the object to a position indistinguishable from where information technology started. In this case the rotation is past 180º, so n=2. A plane of symmetry divides the object in such a style that the points on i side of the airplane are equivalent to the points on the other side past reflection through the plane. In addition to the point of symmetry noted before, (E)-1,two-dichloroethene also has a aeroplane of symmetry (the aeroplane defined by the six atoms), and a C2 axis, passing through the center perpendicular to the plane.
The existence of a reflective symmetry element (a point or airplane of symmetry) is sufficient to assure that the object having that element is achiral .
Chiral objects, therefore, do not have any reflective symmetry elements, but may have rotational symmetry axes, since these elements practice non require reflection to operate. In add-on to the chiral vs achiral distinction, there are two other terms often used to refer to the symmetry of an object. These are:
(i) Dissymmetry : The absence of reflective symmetry elements. All dissymmetric objects are chiral.
(two) Asymmetry : The absence of all symmetry elements. All disproportionate objects are chiral.
Some examples of symmetry elements in simple molecules may be examined past Clicking Here.
Enantiomorphism
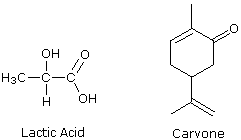 As chemists studied organic compounds isolated from plants and animals, a new and subtle type of configurational stereoisomerism was discovered. For example, lactic acid ( a C3H6O3 carboxylic acid) was establish in sour milk equally well equally in the blood and muscle fluids of animals. The physical properties of this simple chemical compound were identical, regardless of the source (m.p, 53 ºC & pKa 3.lxxx), merely at that place was testify that the physiological beliefs of the compound from the two sources was not the same. Another natural product, the fragrant CtenHxivO ketone carvone, was isolated from both spearmint and caraway. Again, all the physical backdrop of carvone from these two sources seemed to exist identical (b.p. 230 ºC), but the odors of the 2 carvones were different and reflected their source. Other examples of this kind were encountered, and suspicions of a subtle kind of stereoisomerism were confirmed by the dissimilar interaction these compounds displayed with airplane polarized low-cal. We at present know that this configurational stereoisomerism is due to different correct and left-handed forms that certain structures may prefer, in much the same way that a screw may have right or left-handed threads but the aforementioned overall size and shape. Isomeric pairs of this kind are termed enantiomers (from the Greek enantion significant opposite).
As chemists studied organic compounds isolated from plants and animals, a new and subtle type of configurational stereoisomerism was discovered. For example, lactic acid ( a C3H6O3 carboxylic acid) was establish in sour milk equally well equally in the blood and muscle fluids of animals. The physical properties of this simple chemical compound were identical, regardless of the source (m.p, 53 ºC & pKa 3.lxxx), merely at that place was testify that the physiological beliefs of the compound from the two sources was not the same. Another natural product, the fragrant CtenHxivO ketone carvone, was isolated from both spearmint and caraway. Again, all the physical backdrop of carvone from these two sources seemed to exist identical (b.p. 230 ºC), but the odors of the 2 carvones were different and reflected their source. Other examples of this kind were encountered, and suspicions of a subtle kind of stereoisomerism were confirmed by the dissimilar interaction these compounds displayed with airplane polarized low-cal. We at present know that this configurational stereoisomerism is due to different correct and left-handed forms that certain structures may prefer, in much the same way that a screw may have right or left-handed threads but the aforementioned overall size and shape. Isomeric pairs of this kind are termed enantiomers (from the Greek enantion significant opposite).
A consideration of the chirality of molecular configurations explains the curious stereoisomerism observed for lactic acid, carvone and a multitude of other organic compounds. Tetravalent carbons accept a tetrahedral configuration. If all 4 substituent groups are the aforementioned, as in methyl hydride or tetrachloromethane, the configuration is that of a highly symmetric regular tetrahedron. A regular tetrahedron has six planes of symmetry and vii symmetry axes (iv Ciii & three C2) and is, of course, achiral. Examples of these axes and planes were noted above, and may be examined more fully by clicking on the methane formula drawn below.
If one of the carbon substituents is unlike from the other three, the degree of symmetry is lowered to a C3 axis and 3 planes of symmetry, but the configuration remains achiral. The tetrahedral configuration in such compounds is no longer regular, since bond lengths and bond angles change as the bonded atoms or groups change. Further substitution may reduce the symmetry fifty-fifty more, merely as long as ii of the iv substituents are the same there is always a aeroplane of symmetry that bisects the angle linking those substituents, so these configurations are too achiral.
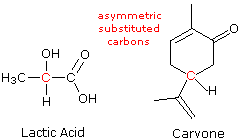 A carbon atom that is bonded to four different atoms or groups loses all symmetry, and is often referred to every bit an asymmetric carbon. The configuration of such a tetrahedral unit is chiral, and the construction may exist in either a right-handed configuration or a left-handed configuration (one the mirror image of the other). This blazon of configurational stereoisomerism is termed enantiomorphism, and the not-identical, mirror-paradigm pair of stereoisomers that outcome are called enantiomers. The structural formulas of lactic acid and carvone are drawn on the correct with the asymmetric carbon colored red. Consequently, we expect, and notice, these compounds to exist equally pairs of enantiomers. The presence of a single asymmetrically substituted carbon atom in a molecule is sufficient to render the whole configuration chiral, and modern terminology refers to such asymmetric (or dissymmetric) groupings as chiral centers. About of the chiral centers we shall discuss are asymmetric carbon atoms, simply it should be recognized that other tetrahedral or pyramidal atoms may become chiral centers if accordingly substituted. When more than one chiral center is present in a molecular structure, care must exist taken to analyze their relationship before concluding that a specific molecular configuration is chiral or achiral. This aspect of stereoisomerism will be treated subsequently.
A carbon atom that is bonded to four different atoms or groups loses all symmetry, and is often referred to every bit an asymmetric carbon. The configuration of such a tetrahedral unit is chiral, and the construction may exist in either a right-handed configuration or a left-handed configuration (one the mirror image of the other). This blazon of configurational stereoisomerism is termed enantiomorphism, and the not-identical, mirror-paradigm pair of stereoisomers that outcome are called enantiomers. The structural formulas of lactic acid and carvone are drawn on the correct with the asymmetric carbon colored red. Consequently, we expect, and notice, these compounds to exist equally pairs of enantiomers. The presence of a single asymmetrically substituted carbon atom in a molecule is sufficient to render the whole configuration chiral, and modern terminology refers to such asymmetric (or dissymmetric) groupings as chiral centers. About of the chiral centers we shall discuss are asymmetric carbon atoms, simply it should be recognized that other tetrahedral or pyramidal atoms may become chiral centers if accordingly substituted. When more than one chiral center is present in a molecular structure, care must exist taken to analyze their relationship before concluding that a specific molecular configuration is chiral or achiral. This aspect of stereoisomerism will be treated subsequently.
The identity or non-identity of mirror-image configurations of some substituted carbons may be examined every bit interactive models by Clicking Here.
A useful outset footstep in examining structural formulas to make up one's mind whether stereoisomers may exist is to identify all stereogenic elements. A stereogenic element is a centre, axis or plane that is a focus of stereoisomerism, such that an interchange of two groups attached to this characteristic leads to a stereoisomer. Stereogenic elements may exist chiral or achiral. The most mutual chiral stereogenic center is the asymmetric carbon; interchanging any two substituent groups converts one enantiomer to the other. However, intendance must be taken when evaluating bridged structures in which bridgehead carbons are asymmetric. This caveat will be illustrated by Clicking Hither.
Alkenes having two different groups on each double bond carbon (e.g. abC=Cab) found an achiral stereogenic element, since interchanging substituents at one of the carbons changes the cis/trans configuration of the double bond. Chiral stereogenic axes or planes may be also be present in a molecular configuration, as in the instance of allenes, but these are less common than chiral centers and will not be discussed hither.
For additional information about allenes Click Here.
Structural formulas for eight organic compounds are displayed in the frame below. Some of these structures are chiral and some are achiral. First, attempt to identify all chiral stereogenic centers. Formulas having no chiral centers are necessarily achiral. Formulas having one chiral center are ever chiral; and if two or more chiral centers are present in a given structure it is likely to be chiral, simply in special cases, to exist discussed after, may be achiral. Once you lot have made your selections of chiral centers, bank check them by pressing the "Prove Chiral Centers" button. The chiral centers will be identified by crimson dots.
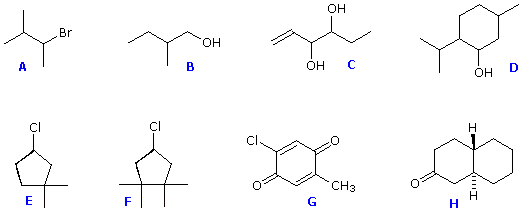
Structures F and G are achiral. The former has a aeroplane of symmetry passing through the chlorine atom and bisecting the reverse carbon-carbon bond. The like construction of compound E does non have such a symmetry plane, and the carbon bonded to the chlorine is a chiral centre (the two ring segments connecting this carbon are not identical). Structure G is essentially flat. All the carbons except that of the methyl group are sp2 hybridized, and therefore trigonal-planar in configuration. Compounds C, D & H have more than 1 chiral heart, and are also chiral. Call up, all chiral structures may exist as a pair of enantiomers. Other configurational stereoisomers are possible if more than than one stereogenic center is nowadays in a structure.
Polarimetry
Optical Activity
Identifying and distinguishing enantiomers is inherently difficult, since their concrete and chemical properties are largely identical. Fortunately, a nearly two hundred yr old discovery past the French physicist Jean-Baptiste Biot has made this task much easier. This discovery disclosed that the correct- and left-handed enantiomers of a chiral compound perturb plane-polarized low-cal in opposite ways. This perturbation is unique to chiral molecules, and has been termed optical activity.
Airplane-polarized lite is created by passing ordinary light through a polarizing device, which may be as simple as a lens taken from polarizing sun-glasses. Such devices transmit selectively just that component of a calorie-free beam having electrical and magnetic field vectors oscillating in a single plane. The plane of polarization can exist determined by an musical instrument called a polarimeter, shown in the diagram below.
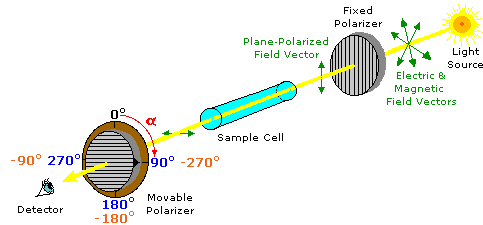
Monochromatic (unmarried wavelength) light, is polarized by a stock-still polarizer next to the calorie-free source. A sample jail cell holder is located in line with the light beam, followed by a movable polarizer (the analyzer) and an eyepiece through which the light intensity tin can be observed. In modern instruments an electronic light detector takes the place of the human eye. In the absence of a sample, the light intensity at the detector is at a maximum when the 2nd (movable) polarizer is ready parallel to the first polarizer (α = 0º). If the analyzer is turned 90º to the plane of initial polarization, all the calorie-free will be blocked from reaching the detector.
Chemists employ polarimeters to investigate the influence of compounds (in the sample jail cell) on airplane polarized low-cal. Samples composed simply of achiral molecules (e.g. h2o or hexane), have no effect on the polarized lite axle. However, if a single enantiomer is examined (all sample molecules being right-handed, or all being left-handed), the plane of polarization is rotated in either a clockwise (positive) or counter-clockwise (negative) direction, and the analyzer must be turned an appropriate matching angle, α, if total lite intensity is to attain the detector. In the above analogy, the sample has rotated the polarization plane clockwise by +90º, and the analyzer has been turned this amount to permit maximum light transmission.
The observed rotations (α) of enantiomers are opposite in direction. One enantiomer will rotate polarized light in a clockwise direction, termed dextrorotatory or (+), and its mirror-image partner in a counter-clockwise fashion, termed levorotatory or (–). The prefixes dextro and levo come from the Latin dexter, meaning right, and laevus, for left, and are abbreviated d and 50 respectively. If equal quantities of each enantiomer are examined , using the aforementioned sample cell, so the magnitude of the rotations will exist the same, with one beingness positive and the other negative. To be absolutely certain whether an observed rotation is positive or negative it is often necessary to brand a second measurement using a unlike amount or concentration of the sample. In the above illustration, for case, α might be –90º or +270º rather than +90º. If the sample concentration is reduced by 10%, then the positive rotation would change to +81º (or +243º) while the negative rotation would modify to –81º, and the right α would be identified unambiguously.
Since information technology is non always possible to obtain or utilize samples of exactly the same size, the observed rotation is usually corrected to recoup for variations in sample quantity and prison cell length. Thus information technology is mutual practise to convert the observed rotation, α, to a specific rotation, [α], by the following formula:
| Specific Rotation = | 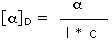 | where l = cell length in dm, c = concentration in g/ml | |
| D is the 589 nm light from a sodium lamp |
Compounds that rotate the plane of polarized calorie-free are termed optically active. Each enantiomer of a stereoisomeric pair is optically agile and has an equal but opposite-in-sign specific rotation. Specific rotations are useful in that they are experimentally determined constants that characterize and identify pure enantiomers. For example, the lactic acid and carvone enantiomers discussed before have the following specific rotations.
| Carvone from caraway: [α]D = +62.5º | this isomer may exist referred to equally (+)-carvone or d-carvone | |
| Carvone from spearmint: [α]D = –62.5º | this isomer may be referred to as (–)-carvone or l-carvone | |
| Lactic acid from musculus tissue: [α]D = +ii.5º | this isomer may be referred to as (+)-lactic acrid or d-lactic acrid | |
| Lactic acrid from sour milk: [α]D = –two.5º | this isomer may be referred to as (–)-lactic acid or l-lactic acid |
A fifty:50 mixture of enantiomers has no observable optical activeness. Such mixtures are called racemates or racemic modifications, and are designated (�). When chiral compounds are created from achiral compounds, the products are racemic unless a single enantiomer of a chiral co-reactant or catalyst is involved in the reaction. The addition of HBr to either cis- or trans-ii-butene is an example of racemic product formation (the chiral center is colored ruby-red in the post-obit equation).
| CH3CH=CHCH3 + HBr |  | (�) CHiiiCHii CHBrCHthree |
Chiral organic compounds isolated from living organisms are commonly optically agile, indicating that one of the enantiomers predominates (frequently it is the only isomer present). This is a event of the action of chiral catalysts we call enzymes, and reflects the inherently chiral nature of life itself. Chiral synthetic compounds, on the other manus, are commonly racemates, unless they have been prepared from enantiomerically pure starting materials.
There are two ways in which the condition of a chiral substance may be inverse:
1. A racemate may exist separated into its component enantiomers. This process is called resolution .
2. A pure enantiomer may exist transformed into its racemate. This process is called racemization .
Designating the Configuration of Chiral Centers
Although enantiomers may be identified past their feature specific rotations, the assignment of a unique configuration to each has not yet been discussed. Nosotros have referred to the mirror-prototype configurations of enantiomers as "right-handed" and "left-handed", simply deciding which is which is not a trivial chore. An early process assigned a D prefix to enantiomers chemically related to a right-handed reference chemical compound and a L prefix to a similarly related left-handed group of enantiomers. Although this notation is still applied to carbohydrates and amino acids, information technology required chemic transformations to establish group relationships, and proved to be ambiguous in its general application. A concluding solution to the vexing problem of configuration assignment was devised by three European chemists: R. Due south. Cahn, C. Grand. Ingold and V. Prelog. The resulting nomenclature system is sometimes chosen the CIP arrangement or the R-S organization.
In the CIP system of nomenclature, each chiral middle in a molecule is assigned a prefix (R or South), according to whether its configuration is right- or left-handed. No chemical reactions or interrelationship are required for this assignment. The symbol R comes from the Latin rectus for right, and L from the Latin sinister for left. The assignment of these prefixes depends on the application of two rules: The Sequence Rule and The Viewing Rule.
The sequence rule is the same equally that used for assigning E-Z prefixes to double bond stereoisomers. Since near of the chiral stereogenic centers we shall encounter are disproportionate carbons, all four different substituents must be ordered in this fashion.
The Sequence Rule for Assignment of Configurations to Chiral Centers
Assign sequence priorities to the four substituents by looking at the atoms attached straight to the chiral eye.
1. The higher the atomic number of the firsthand substituent atom, the higher the priority.
For example, H– < C– < Northward– < O– < Cl–. (Different isotopes of the same element are assigned a priority according to their diminutive mass.)
2. If two substituents take the same immediate substituent cantlet,
evaluate atoms progressively further away from the chiral center until a divergence is found.
For example, CHiii– < CiiH5– < ClCHii– < BrCH2– < CH3O–.
3. If double or triple bonded groups are encountered as substituents, they are treated equally an equivalent set of single-bonded atoms.
For instance, C2Hv– < CH2=CH– < HC≡C–
One time the relative priorities of the four substituents have been determined, the chiral middle must be viewed from the side opposite the lowest priority group. If nosotros number the substituent groups from 1 to 4, with 1 being the highest and four the lowest in priority sequence, the two enantiomeric configurations are shown in the post-obit diagram along with a viewers eye on the side contrary substituent #4.

Remembering the geometric implication of wedge and hatched bonds, an observer (the eye) notes whether a curved arrow drawn from the # 1 position to the # 2 location and so to the # 3 position turns in a clockwise or counter-clockwise manner. If the turn is clockwise, every bit in the case on the right, the configuration is classified R. If it is counter-clockwise, as in the left illustration, the configuration is S. Another way of remembering the viewing rule, is to call up of the asymmetric carbon every bit a steering wheel. The bond to the lowest priority group (# 4) is the steering column, and the other bonds are spokes on the wheel. If the wheel is turned from group # one toward group # two, which in turn moves toward group # 3, this would either negotiate a right turn (R) or a left turn (S). This model is illustrated below for a right-handed turn, and the respective (R)-configurations of lactic acid and carvone are shown to its correct. The stereogenic carbon atom is colored magenta in each instance, and the sequence priorities are shown as low-cal blue numbers. Note that if whatever 2 substituent groups on a stereogenic carbon are exchanged or switched, the configuration changes to its mirror prototype.
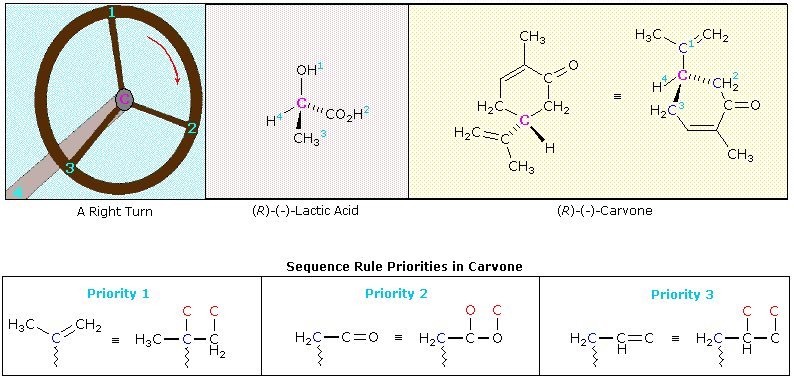
The sequence guild of the substituent groups in lactic acid should be obvious, only the carvone instance requires careful analysis. The hydrogen is clearly the lowest priority substituent, simply the other three groups are all fastened to the stereogenic carbon by bonds to carbon atoms (colored blue here). Two of the immediate substituent species are methylene groups (CH2), and the tertiary is a doubly-bonded carbon. Rule # 3 of the sequence rules allows usa to order these substituents. The carbon-carbon double bond is broken then every bit to give imaginary unmarried-bonded carbon atoms (the phantom atoms are colored red in the equivalent structure). In this form the double bond assumes the priority of a 3º-alkyl group, which is greater than that of a methylene group. To found the sequence priority of the 2 methylene substituents (both are part of the ring), we must move away from the chiral heart until a signal of difference is located. This occurs at the next carbon, which on one side is part of a carbonyl double bond (C=O), and on the other, part of a carbon-carbon double bond. Rule # 3 is again used to evaluate the two cases. The carbonyl group places two oxygens (one phantom) on the adjacent carbon atom, and then this methylene side is ranked ahead of the other.
An interesting feature of the two examples shown here is that the R-configuration in both cases is levorotatory (-) in its optical activity. The mirror-image Southward-configurations are, of grade, dextrorotatory (+). It is of import to remember that there is no simple or obvious human relationship between the R or S designation of a molecular configuration and the experimentally measured specific rotation of the chemical compound it represents. In guild to determine the true or "absolute" configuration of an enantiomer, every bit in the cases of lactic acrid and carvone reported hither, it is necessary either to relate the compound to a known reference structure, or to conduct a rather complex X-ray analysis on a single crystal of the sample.
The configurations of lactic acid and carvone enantiomers may exist examined equally interactive models by Clicking Here.
| The module on the right provides examples of chiral and achiral molecules for assay. These are displayed equally iii-dimensional structures which may be moved almost and examined from various points of view. By using this resources the reader'south understanding of configurational note may be tested. This visualization makes use of the Jmol applet. With some browsers information technology may be necessary to click a button twice for action. |
| |||||||
Compounds Having Two or More than Chiral Centers
The Chinese shrub Ma Huang (Ephedra vulgaris) contains two physiologically active compounds ephedrine and pseudoephedrine. Both compounds are stereoisomers of ii-methylamino-1-phenyl-1-propanol, and both are optically active, one being levorotatory and the other dextrorotatory. Since the properties of these compounds (see below) are significantly dissimilar, they cannot be enantiomers. How, and so, are nosotros to classify these isomers and others like them?
| Ephedrine from Ma Huang: | thousand.p. 35 - 40 º C, [α]D = –41º, moderate water solubility | [this isomer may exist referred to as (–)-ephedrine] |
| Pseudoephedrine from Ma Huang: | grand.p. 119 º C, [α]D = +52º, relatively insoluble in water | [this isomer may be referred to every bit (+)-pseudoephedrine] |
Since these two compounds are optically active, each must have an enantiomer. Although these missing stereoisomers were not present in the natural source, they have been prepared synthetically and have the expected identical concrete properties and opposite-sign specific rotations with those listed above. The structural formula of 2-methylamino-one-phenylpropanol has two stereogenic carbons (#1 & #2). Each may assume an R or S configuration, so in that location are four stereoisomeric combinations possible. These are shown in the following illustration, together with the assignments that have been made on the footing of chemical interconversions.
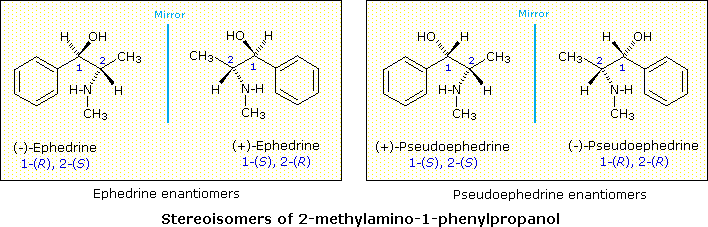
Every bit a general rule, a structure having north chiral centers volition have 2n possible combinations of these centers . Depending on the overall symmetry of the molecular structure, some of these combinations may be identical, only in the absence of such identity, nosotros would expect to find 2n stereoisomers. Some of these stereoisomers volition have enantiomeric relationships, but enantiomers come in pairs, and non-enantiomeric stereoisomers volition therefore be common. We refer to such stereoisomers as diastereomers. In the example above, either of the ephedrine enantiomers has a diastereomeric relationship with either of the pseudoephedrine enantiomers.
For an interesting instance illustrating the stardom betwixt a chiral center and an asymmetric carbon Click Here.
The configurations of ephedrine and pseudoephedrine enantiomers may be examined equally interactive models by Clicking Here.
Stereogenic Nitrogen
A close examination of the ephedrine and pseudoephedrine isomers suggests that another stereogenic centre, the nitrogen, is present. As noted earlier, single-bonded nitrogen is pyramidal in shape, with the not-bonding electron pair pointing to the unoccupied corner of a tetrahedral region. Since the nitrogen in these compounds is bonded to three unlike groups, its configuration is chiral. The non-identical mirror-image configurations are illustrated in the following diagram (the remainder of the molecule is represented by R, and the electron pair is colored yellow). If these configurations were stable, there would be 4 additional stereoisomers of ephedrine and pseudoephedrine. Nevertheless, pyramidal nitrogen is normally not configurationally stable. It apace inverts its configuration (equilibrium arrows) by passing through a planar, sp2-hybridized transition state, leading to a mixture of interconverting R and S configurations. If the nitrogen atom were the only chiral center in the molecule, a 50:50 (racemic) mixture of R and S configurations would exist at equilibrium. If other chiral centers are present, as in the ephedrin isomers, a mixture of diastereomers will event. In any upshot, nitrogen groups such as this, if present in a compound, do not contribute to isolable stereoisomers.
The inversion of pyramidal nitrogen in ammonia may exist examined past clicking on the following diagram.
 |
|---|
Fischer Projection Formulas
The problem of drawing three-dimensional configurations on a two-dimensional surface, such every bit a piece of paper, has been a long-standing concern of chemists. The wedge and hatched line notations we take been using are effective, but can be troublesome when applied to compounds having many chiral centers. Equally part of his Nobel Prize-winning research on carbohydrates, the cracking German chemist Emil Fischer, devised a simple notation that is even so widely used. In a Fischer projection drawing, the four bonds to a chiral carbon brand a cross with the carbon atom at the intersection of the horizontal and vertical lines. The two horizontal bonds are directed toward the viewer (forward of the stereogenic carbon). The two vertical bonds are directed behind the fundamental carbon (away from the viewer). Since this is not the usual manner in which nosotros accept viewed such structures, the following diagram shows how a stereogenic carbon positioned in the common 2-bonds-in-a-airplane orientation ( ten–C–y define the reference plane ) is rotated into the Fischer projection orientation (the far correct formula). When writing Fischer projection formulas it is important to recall these conventions. Since the vertical bonds extend away from the viewer and the horizontal bonds toward the viewer, a Fischer structure may but be turned past 180º within the airplane, thus maintaining this relationship. The structure must not exist flipped over or rotated by 90º.
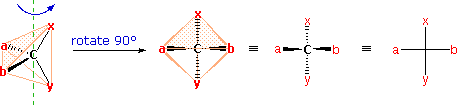
A model showing the above rotation into a Fischer projection may exist examined by Clicking Here.
In the to a higher place diagram, if 10 = CO2H, y = CH3, a = H & b = OH, the resulting formula describes (R)-(–)-lactic acid. The mirror-image formula, where x = CO2H, y = CH3, a = OH & b = H, would, of class, stand for (South)-(+)-lactic acid.
Using the Fischer project notation, the stereoisomers of ii-methylamino-1-phenylpropanol are fatigued in the following style. Annotation that it is customary to set the longest carbon chain as the vertical bond assembly.

The usefulness of this notation to Fischer, in his carbohydrate studies, is evident in the post-obit diagram. At that place are eight stereoisomers of 2,three,iv,5-tetrahydroxypentanal, a group of compounds referred to as the aldopentoses. Since there are three chiral centers in this constitution, we should expect a maximum of 23 stereoisomers. These eight stereoisomers consist of four sets of enantiomers. If the configuration at C-4 is kept constant (R in the examples shown here), the 4 stereoisomers that result will be diastereomers. Fischer formulas for these isomers, which Fischer designated equally the "D"-family, are shown in the diagram. Each of these compounds has an enantiomer, which is a member of the "L"-family so, as expected, there are eight stereoisomers in all. Determining whether a chiral carbon is R or S may seem hard when using Fischer projections, but information technology is actually quite simple. If the lowest priority group (often a hydrogen) is on a vertical bond, the configuration is given directly from the relative positions of the three higher-ranked substituents. If the lowest priority grouping is on a horizontal bond, the positions of the remaining groups give the wrong answer (you lot are in looking at the configuration from the wrong side), so you simply opposite it.
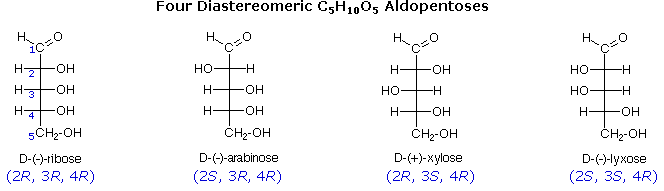
The aldopentose structures fatigued above are all diastereomers. A more selective term, epimer, is used to designate diastereomers that differ in configuration at only one chiral middle. Thus, ribose and arabinose are epimers at C-2, and arabinose and lyxose are epimers at C-3. However, arabinose and xylose are not epimers, since their configurations differ at both C-2 and C-3.
Achiral Diastereomers (Meso-Compounds)
The chiral centers in the preceding examples have all been unlike, one from another. In the instance of 2,iii-dihydroxybutanedioic acid, known as tartaric acid, the two chiral centers have the aforementioned iv substituents and are equivalent. As a consequence, two of the 4 possible stereoisomers of this compound are identical due to a airplane of symmetry, and then there are only three stereoisomeric tartaric acids. Two of these stereoisomers are enantiomers and the third is an achiral diastereomer, called a meso compound. Meso compounds are achiral (optically inactive) diastereomers of chiral stereoisomers. Investigations of isomeric tartaric acid salts, carried out by Louis Pasteur in the mid 19th century, were instrumental in elucidating some of the subtleties of stereochemistry.
Some concrete properties of the isomers of tartaric acid are given in the following table.
| (+)-tartaric acid: | [α]D = +13º | 1000.p. 172 ºC |
| (–)-tartaric acid: | [α]D = –13º | chiliad.p. 172 ºC |
| meso-tartaric acid: | [α]D = 0º | m.p. 140 ºC |
Fischer projection formulas provide a helpful view of the configurational relationships within the structures of these isomers. In the post-obit illustration a mirror line is drawn between formulas that have a mirror-image relationship. In demonstrating the identity of the two meso-chemical compound formulas, remember that a Fischer projection formula may be rotated 180º in the airplane.
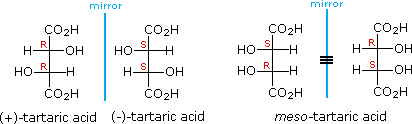
A model of meso-tartaric acrid may be examined past Clicking Hither.
An additional case, consisting of two meso compounds, may be examined by Clicking Here.
Other methods of designating configuration take been proposed. These volition be shown by Clicking Hither.
Resolution of Racemates
Every bit noted earlier, chiral compounds synthesized from achiral starting materials and reagents are generally racemic (i.due east. a 50:fifty mixture of enantiomers). Separation of racemates into their component enantiomers is a process called resolution. Since enantiomers have identical physical properties, such as solubility and melting point, resolution is difficult. Diastereomers, on the other hand, have dissimilar physical properties, and this fact may exist used to reach resolution of racemates. Reaction of a racemate with an enantiomerically pure chiral reagent gives a mixture of diastereomers, which tin can be separated. Reversing the first reaction then leads to the separated enantiomers plus the recovered reagent.
Many kinds of chemic and physical reactions, including salt formation, may be used to achieve the diastereomeric intermediates needed for separation. The post-obit diagram illustrates this general principle by showing how a nut having a right-handed thread (R) could serve as a "reagent" to discriminate and separate a mixture of correct- and left-handed bolts of identical size and weight. Only the two right-handed partners can collaborate to requite a fully-threaded intermediate, and then separation is fairly simple. The resolving moiety, i.eastward. the nut, is so removed, leaving the bolts separated into their correct and left-handed forms. Chemical reactions of enantiomers are ordinarily not then dramatically dissimilar, only a practical distinction is nevertheless possible.
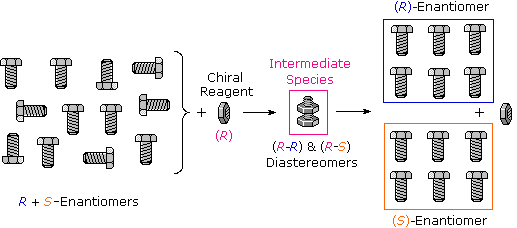
To see how resolution was first achieved, and how new methods have evolved Click Here.
Conformational Enantiomorphism
The Fischer projection formula of meso-tartaric acrid has a airplane of symmetry bisecting the C2–C3 bond, equally shown on the left in the diagram below, and then this structure is clearly achiral. The eclipsed orientation of bonds that is assumed in the Fischer drawing is, yet, an unstable conformation, and we should examine the staggered conformers that undoubtedly make upwardly most of the sample molecules. The 4 structures that are shown to the correct of the Fischer projection consist of the achiral Fischer conformation (A) and three staggered conformers, all displayed in both sawhorse and Newman projections. The second and fourth conformations (B & D) are dissymmetric, and are in fact enantiomeric structures. The third conformer (C) has a center of symmetry and is achiral.
Conformations of meso-Tartaric Acid | ||||
|---|---|---|---|---|
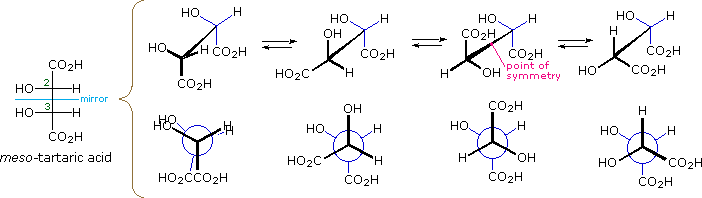 | ||||
| Fischer Projection | A eclipsed, achiral | B staggered, chiral | C staggered, achiral | D staggered, chiral |
Since a meaning proportion of the meso-tartaric acrid molecules in a sample volition have chiral conformations, the achiral properties of the sample (eastward.m. optical inactivity) should not be attributed to the symmetry of the Fischer formula. Equilibria among the various conformations are chop-chop established, and the proportion of each conformer present at equilibrium depends on its relative potential energy (the most stable conformers predominate). Since enantiomers have equal potential energies, they will be present in equal concentration, thus canceling their macroscopic optical activity and other chiral behavior. Merely put, any chiral species that are nowadays are racemic.
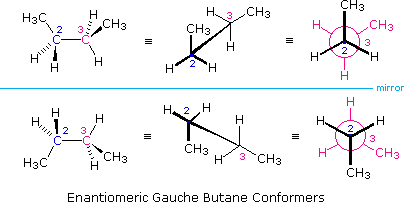
It is interesting to notation that chiral conformations are nowadays in most conformationally mobile compounds, even in the absence of any chiral centers. The gauche conformers of butane, for example, are chiral and are present in equal concentration in whatever sample of this hydrocarbon. The following illustration shows the enantiomeric relationship of these conformers, which are an instance of a chiral axis rather than a chiral heart.
Substituted biphenyls may be as isolable enantiomers. This will be demonstrated by Clicking Here.
Stereoisomerism in Disubstituted Cyclohexanes
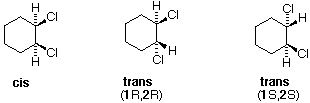
The distinction betwixt configurational stereoisomers and the conformers they may assume is well-illustrated past the disubstituted cyclohexanes. The following discussion uses the various isomers of dichlorocyclohexane as examples. The one,1-dichloro isomer is omitted because it is an unexceptional constitutional isomer of the others, and has no centers of chirality (asymmetric carbon atoms). The i,2- and 1,3-dichlorocyclohexanes each have two centers of chirality, bearing the same set of substituents. The cis & trans-ane,iv-dichlorocyclohexanes do not have any chiral centers, since the 2 ring groups on the substituted carbons are identical.
There are iii configurational isomers of 1,2-dichlorocyclohexane and three configurational isomers of 1,3-dichlorocyclohexane. These are shown in the following table.
| The 1,2-Dichlorocyclohexanes | The i,three-Dichlorocyclohexanes |
|---|---|
 | 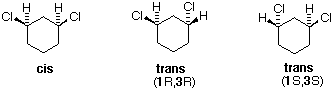 |
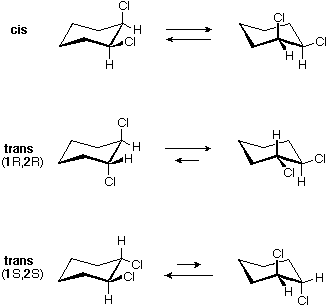 | 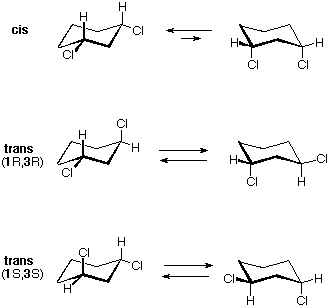 |
All the i,2-dichloro isomers are constitutional isomers of the ane,3-dichloro isomers. In each category (1,2- & 1,3-), the (R,R)-trans isomer and the (S,Southward)-trans isomer are enantiomers. The cis isomer is a diastereomer of the trans isomers. Finally, all of these isomers may exist equally a mixture of two (or more) conformational isomers, equally shown in the table.
The chair conformer of the cis 1,ii-dichloro isomer is chiral. It exists every bit a 50:l mixture of enantiomeric conformations, which interconvert then rapidly they cannot be resolved (ie. separated). Since the cis isomer has ii centers of chirality (asymmetric carbons) and is optically inactive, it is a meso-compound. The corresponding trans isomers also exist as apace interconverting chiral conformations. The diequatorial conformer predominates in each case, the (R,R) conformations being mirror images of the (S,S) conformations. All these conformations are diastereomeric with the cis conformations.
The diequatorial chair conformer of the cis 1,3-dichloro isomer is achiral. It is the major component of a fast equilibrium with the diaxial conformer, which is also achiral. This isomer is too a meso compound. The respective trans isomers too undergo a rapid conformational interconversion. For these isomers, however, this interconversion produces an identical conformer, and so each enantiomer (R,R) and (S,Southward) has predominately a single chiral conformation. These enantiomeric conformations are diastereomeric with the cis conformations.
The 1,4-dichlorocyclohexanes may exist every bit cis or trans stereoisomers. Both are achiral, since the disubstituted 6-membered band has a plane of symmetry. These isomers are diastereomers of each other, and are ramble isomers of the i,2- and 1,three- isomers.
The 1,4-Dichlorocyclohexanes
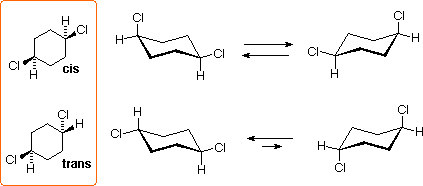
All the chair conformers of these isomers are achiral, and the diequatorial conformer of the trans isomer is the predominate species at equilibrium.
Advanced Topics |
|---|
Recognizing the structural equivalence or not-equivalence of groups is essential for stereochemical assay.
For examples of this analysis, and the classification of groups every bit homotopic or heterotopic Click Here.
Stereogenic elements may be chiral or achiral. Examples will exist displayed past Clicking Here.
| Practise Issues |
|---|
The kickoff v problems enquire you to identify equivalent groups of atoms, symmetry elements, stereogenic centers and the presence or absenteeism of chirality. Office two of the 4th problem also requires the application of R/S nomenclature. The nomenclature terminology and nomenclature of stereoisomers is examined in the next ii problems, followed by a question apropos the relationship of isomeric pairs. Designation of CIP names is the bailiwick of the adjacent four bug. Products from stereoselective reactions are examined in the next problem, and a review of cyclohexane conformational terminology is the subject of the last two problems.
Full general Summary of Isomerism and Molecular Descriptors
Methods of Describing Molecules with Increasing Refinement
-
Composition
The number and kinds of atoms that make upwards a molecule. This information is supplied by a molecular formula. -
Constitution
The bonding pattern of the atoms of a molecule (ie. which atoms are connected to which other atoms and by what kind of bonds). Dissimilar bonding constitutions are interconverted only by breaking and reforming covalent bonds. This information is supplied by a structural formula, and is implicit in the IUPAC proper noun. -
Configuration
The permanent spatial relationship of the atoms of a molecule to each other. Different configurations are interconverted but by breaking and reforming covalent bonds. This data is given in a stereo-formula, and is likewise provided by a prefix to the IUPAC name (eg. cis & trans). -
Conformation
The variable spatial orientation of the atoms of a molecule to each other that occurs past rotation or twisting of bonds. Dissimilar conformations are interconverted without breaking covalent bonds. This information is supplied by conformational formulas, and as well past nomenclature terms (eg. gauche & anti).
Relationship of Ramble and Stereoisomers
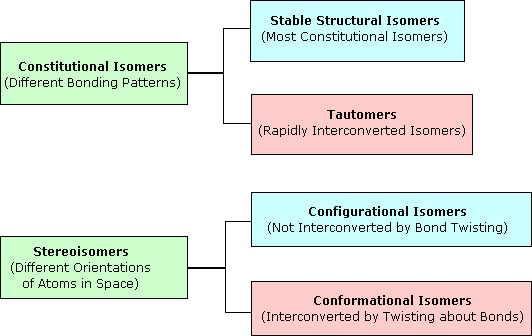
Relationships of Stereoisomers

| Render to Table of Contents |
|---|
Source: https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/chapt11.htm
0 Response to "How Do You Know if a Mokecuel Is Chiral or Achiral"
Postar um comentário